 But this number physically corresponds to the actual weight of an atom. 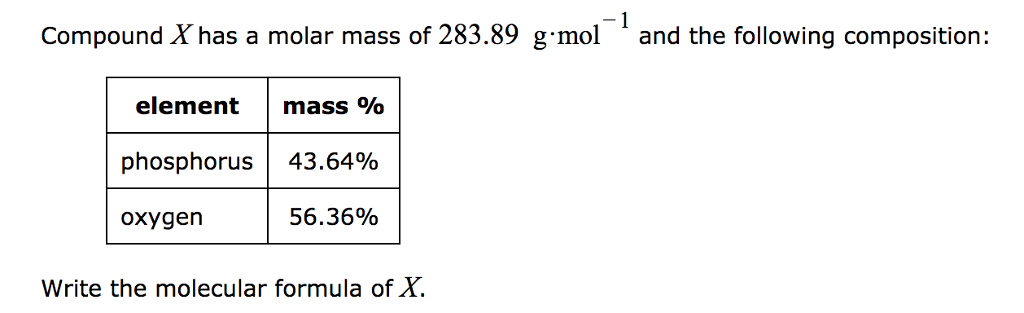
× Relative molecular mass of carbon dioxideĪtomic weights ( M) of atoms are related to the sum of the number of protons and neutrons. Mass of carbon dime = Number of moles of carbon dioxide In order to find the mass of carbon dioxide gas occupying 100 cm 3 at 273 K and 101.3 kPa, use is made of the fact that 100 cm 3 of CO 2 is equivalent to 0.00446 moles. = 1 224 or 0.00446 moles of carbon dioxide. Thus, 100 cm 3 of CO 2 are equivalent to 1 2240 × 100 moles of CO 2 Thus, 22400 cm 3 of CO 2 are equivalent to 1 mole of CO 2.Īnd 1 cm 3 of CO 2 is equivalent to 1 22400 moles of CO 2. Use is made of the above definition that at 101.3 kPa and 273 K, 22400 cm 3 of any gas is the volume of 1 mole of the gas. For example, to find the number of moles of carbon dioxide gas which are contained in 100 cm 3 of the gas measured at 273 K and 101.3 kPa. 
Using the molar volume definition, if the volume of a gas is known, the number of moles and hence the mass of the gas can be determined. Volumes of gases are easier to measure than masses. When applied to gases, the molar volume of any gas is defined as occupying 22.4 dm 3 at a temperature of 273 K and pressure 101.3 kPa (atmospheric pressure). That is, the mass of sodium hydroxide required is 16 g. Mass of NaOH = number of moles × relative molecular of NaOH mass of NaOH In order to find the mass of sodium hydroxide required to make 200 cm 3 of 2 M solution:Ģ00 cm 3 of a 2 M solution requi× 200 moles.Ġ.4 moles of NaOH has a mass found by the equation: That is, the number of moles of sodium hydroxide is 0.4. Thus, 1 cm 3 of the solution contamoles of NaOH and 200 cm 3 of the solution conta× 200 moles of NaOH. For example, to find how many moles of sodium hydroxide, NaOH, are contained in 200 cm 3 of a 2 M, (2 molar), solution:ġ000 cm 3 of the solution contains 2 moles of NaOH This means that if the concentration of the solution is known in moles per dm 3, the number of moles in any volume of solution can be determined. When applied to solutions, a 1 molar, (1 M), solution is one in which 1 mole of a solute is dissolved in a solvent in order that the volume of the solution is 1000 cm 3 ( 1 dm 3 or 1 litre). For many purposes the relative atomic masses are rounded up to the nearest whole number except for chlorine and copper which are 35.5 and 63.5 respectively. 
For example, calcium carbonate contains calcium, carbon and oxygen in the ratio 1:1, 3 (i.e. When applied to molecules, one mole of molecules is the relative molecular mass of that molecule, which is the summation of the individual relative atomic masses of the constituent atoms. Thus, one mole of sodium weighs 23.0 g or one tenth of a mole of sodium weighs 2.3 g. The word ‘mole’ has been adopted to represent the Avogadro number of atoms of an element, that is, the relative atomic mass of an element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
